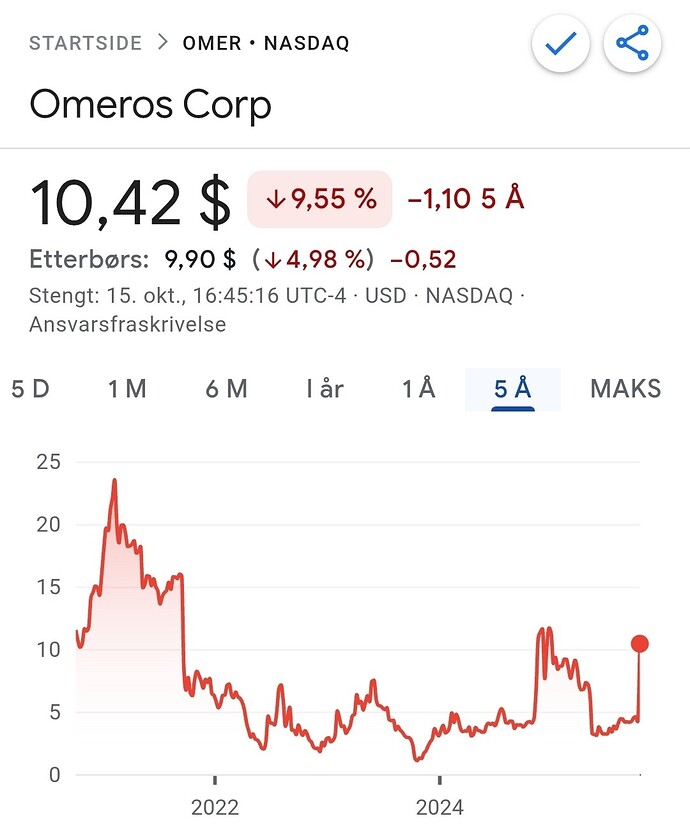
Mulig opptrenden er i gang savepig. Vil nok bare fortsette å gå nesten måned på forventninger om BLA innsendelse. Etter det er det iallefall ingen grunn til å selge.
Du er blitt en ekte Omoron!
Omer er en gammel flamme 
Blir fort dobling om man får ordnet på pass through av Omidria, og hvis ikke så burde det på det nærmeste være priset inn. Gir ellers ikke mye mening at aksjekursen er her nede gitt BLA filing og sannsynlig priority review i nærmeste fremtid.
De hadde jo nettopp emi… Det kommer nok en flere ja, men forhåpentligvis på endel høyere kurser neste år. Vi har en rekke potensielt gode nyheter forran oss som starter med narso data imorgen.
https://investor.omeros.com/news-releases/news-release-details/omeros-reports-final-efficacy-and-safety-data-narsoplimab Råsterke endelige data. Så at narso blir godkjent for HSCT-TMA er gitt. Men BLA er ikke fullstendig sendt inn enda
Hører på webcast. BLA er >70% published
Omers narsoplimab vise god effekt på veldig syke covid pasienter selv om det er en liten sample. 7 av 7 som fikk mekanisk ventilering ble frisk og utskrevet. I artikkelen står det 6, men de har behandlet en pasient i ettertid også og har flere under behandling nå såvidt jeg har skjønt. Forventer news om at det innledes en studie eller at narsoplimab klargjøres for bruk i behandling.
https://investor.omeros.com/static-files/76b8bb70-b7e4-44d1-ad10-fd7136918449?fbclid=IwAR13xWnHHz1UNFngM8m5MZ7eP8KH5W5tIhUupcEq2pADKZFYSYirHJIAfnk Spennede slides fra presentasjon igår.
Approximately 50 patients have been dosed with narsoplimab across the two EIS
Marked improvement seen in narsoplimab-treated patients in these studies
Forventer det kommer noe covid news her snart
Government contract awards and clinical trials now ticker-tagged
12 MINUTES AGO | 3:00 P.M. CET | BUSINESSWIRE
Omeros Submits its Biologics License Application to U.S. FDA for Narsoplimab in HSCT-TMA
Stocks mentioned: OMER
Share
Listen
Save
SEATTLE–(BUSINESS WIRE)–Omeros Corporation (Nasdaq: OMER) announced that it has completed the rolling submission of its Biologics License Application (BLA) to the U.S. Food and Drug Administration (FDA) for narsoplimab for the treatment of hematopoietic stem cell transplant-associated thrombotic microangiopathy (HSCT-TMA). Narsoplimab targets mannan-binding lectin-associated serine protease-2 (MASP-2), the effector enzyme of the lectin pathway of complement, and has received breakthrough therapy designation and orphan drug designation from FDA for HSCT-TMA.
This final portion of the rolling BLA submission, comprised of the BLA’s clinical sections, follows the previously submitted chemistry, manufacturing and controls (CMC) and nonclinical sections, which are under review by FDA. The clinical sections are based on previously reported results of the pivotal trial of narsoplimab in HSCT-TMA in which the drug met its primary endpoint and demonstrated similarly strong response across its secondary endpoints.
Omeros has requested priority review for the BLA, and applicable regulations provide FDA up to 60 days to determine the filing and review designation for the application.
Statistisk er det over 90% sjans for markedsgodkjennig når man har sendt inn en BLA, men denne sykdommen har inge behandling og resultatene er veldig gode så jeg vil tippe det er bortimot 100% sikkert at de får priority review og godkjenning.
Omeros (NASDAQ:OMER) +24.7%post-market after a rule from the Centers for Medicare & Medicaid Services showed the company’s Omidria qualifies as a non-opioid pain management drug that functions as a surgical supply.
According to Bloomberg, the ruling means Omidria will be excluded from packaging under the ambulatory surgical center payment system for 2021.
Så ble det priority review og. Boom.
Shit is getting real